Pharmaceutical Safety Assessment Market 2026 Strengthening Compliance And Risk Evaluation

The Business Research Company’s Pharmaceutical Safety Assessment Global Market Report 2026 – Market Size, Trends, And Forecast 2026-2035
The Business Research Company’s Pharmaceutical Safety Assessment Global Market Report 2026 – Market Size, Trends, And Forecast 2026-2035
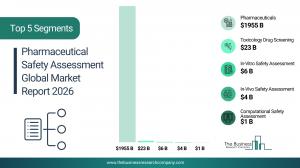
LONDON, GREATER LONDON, UNITED KINGDOM, March 10, 2026 /EINPresswire.com/ -- Pharmaceutical Safety Assessment market to surpass $17 billion in 2030. In comparison, the Toxicology Drug Screening market, which is considered as its parent market, is expected to be approximately $33 billion by 2030, with Pharmaceutical Safety Assessment to represent around 52% of the parent market. Within the broader Pharmaceuticals industry, which is expected to be $1,855 billion by 2030, the Pharmaceutical Safety Assessment market is estimated to account for nearly 0.9% of the total market value.
Which Will Be The Biggest Region In The Pharmaceutical Safety Assessment Market In 2030
North America will be the largest region in the pharmaceutical safety assessment market in 2030, valued at $6 billion. The market is expected to grow from $4 billion in 2025 at a compound annual growth rate (CAGR) of 7%. The strong growth can be attributed to increasing pharmaceutical and biotechnology R&D expenditure, stringent regulatory requirements for drug safety evaluation, rising clinical trial activities, strong presence of contract research organizations, growing development of biologics and specialty drugs, and continuous adoption of advanced toxicology and risk assessment technologies across the United States and Canada.
Which Will Be The Largest Country In The Global Pharmaceutical Safety Assessment Market In 2030?
The USA will be the largest country in the pharmaceutical safety assessment market in 2030, valued at $5 billion. The market is expected to grow from $3 billion in 2025 at a compound annual growth rate (CAGR) of 7%. The strong growth can be attributed to expanding drug development pipelines, increasing outsourcing of toxicology and safety studies to contract research organizations, rising regulatory requirements for preclinical and clinical safety evaluation, growing biologics and biosimilars development, increasing adoption of advanced in vitro and computational toxicology models, and continuous investments in pharmacovigilance and risk assessment infrastructure across the country.
Request A Free Sample Of The Pharmaceutical Safety Assessment Market Report:
https://www.thebusinessresearchcompany.com/sample_request?id=29147&type=smp&utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Mar_PR
What Will Be Largest Segment In The Pharmaceutical Safety Assessment Market In 2030?
The pharmaceutical safety assessment market is segmented by type into in-vitro safety assessment, in-vivo safety assessment, and computational safety assessment. in-vitro safety assessment market will be the largest segment of the pharmaceutical safety assessment market segmented by type, accounting for 55% or $9 billion of the total in 2030. The in-vitro safety assessment market will be supported by the increasing adoption of cell-based assays and organ-on-chip technologies, rising demand for high-throughput screening methods, stringent regulatory emphasis on reducing animal testing, growing biologics pipelines, and continuous advancements in predictive toxicology models.
The pharmaceutical safety assessment market is segmented by drug type into biological drugs and chemical drugs.
The pharmaceutical safety assessment market is segmented by phase into preclinical, clinical (phase I, II, III), and post-market surveillance (phase IV).
The pharmaceutical safety assessment market is segmented by end user into pharmaceutical companies, biotechnology companies, medical device companies, academic and research institutions, and contract research organizations (CROs).
What Is The Expected CAGR For The Pharmaceutical Safety Assessment Market Leading Up To 2030?
The expected CAGR for the pharmaceutical safety assessment market leading up to 2030 is 8%.
What Will Be The Growth Driving Factors In The Global Pharmaceutical Safety Assessment Market In The Forecast Period?
The rapid growth of the global pharmaceutical safety assessment market leading up to 2030 will be driven by the following key factors that are expected to accelerate drug and biologics development, strengthen regulatory compliance requirements, expand chronic disease treatment pipelines, and enhance comprehensive safety evaluation across the pharmaceutical product lifecycle.
Growing Demand For New Drugs And Biologics - The growing demand for new drugs and biologics is expected to become a key growth driver for the pharmaceutical safety assessment market by 2030. The increasing demand for novel drugs and biologics is propelled by the rising prevalence of chronic and infectious diseases, expanding aging populations, and the growing complexity of healthcare needs worldwide. Progress in biotechnology has significantly accelerated biologics development, thereby increasing the need for comprehensive safety assessments. Furthermore, the advancement of personalized medicine and rare disease treatments is driving innovation, requiring thorough safety evaluations to comply with stringent regulatory requirements. As a result, the growing demand for new drugs and biologics is anticipated to contributing to 3.0% annual growth in the market.
Rising Prevalence Of Chronic Diseases Globally - The rising prevalence of chronic diseases globally is expected to emerge as a major factor driving the expansion of the pharmaceutical safety assessment market by 2030. The worldwide increase in chronic conditions such as diabetes, cancer, and cardiovascular diseases is fueling demand for sustained and innovative therapeutic solutions. This growing need accelerates the requirement for comprehensive pharmaceutical safety assessments to ensure drug effectiveness while reducing potential long-term adverse effects. Since chronic illnesses often require ongoing treatment, continuous safety evaluation and monitoring remain critical throughout the entire drug lifecycle. Consequently, the rising prevalence of chronic diseases globally is projected to contribute to around 2.9% annual growth in the market.
Rise In Pharmaceutical R&D Spending And Drug Development Pipelines - The rise in pharmaceutical R&D spending and drug development pipelines is expected to act as a key growth catalyst for the pharmaceutical safety assessment market by 2030. Rising investment in pharmaceutical research and development, along with expanding drug pipelines, highlights the global drive toward innovative therapies and accelerated approval processes. As a growing number of drug candidates progress through preclinical and clinical stages, the need for detailed safety assessments intensifies. At the same time, increasingly stringent regulatory requirements make comprehensive safety evaluation an essential component of the drug development process. Therefore, the rise in pharmaceutical R&D spending and drug development pipelines is projected to contribute to approximately 2.5% annual growth in the market.
Access The Detailed Pharmaceutical Safety Assessment Market Report Here:
https://www.thebusinessresearchcompany.com/report/pharmaceutical-safety-assessment-global-market-report?utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Mar_PR
What Are The Key Growth Opportunities In Pharmaceutical Safety Assessment Market In 2030?
The most significant growth opportunities are anticipated in the in-vitro safety assessment market, the in-vivo safety assessment market, and the computational safety assessment market. Collectively, these segments are projected to contribute over $6 billion in market value by 2030, driven by expanding drug development pipelines, rising outsourcing of toxicology studies to contract research organizations, increasing adoption of advanced cell-based and organ-on-chip models, growing integration of artificial intelligence and predictive toxicology tools, and tightening regulatory requirements for comprehensive safety evaluation.
The in-vitro safety assessment market is projected to grow by $3 billion, in-vivo safety assessment market by $2 billion, and the computational safety assessment market by $1 billion over the next five years from 2025 to 2030.
Learn More About The Business Research Company
The Business Research Company (www.thebusinessresearchcompany.com) is a leading market intelligence firm renowned for its expertise in company, market, and consumer research. We have published over 17,500 reports across 27 industries and 60+ geographies. Our research is powered by 1,500,000 datasets, extensive secondary research, and exclusive insights from interviews with industry leaders.
We provide continuous and custom research services, offering a range of specialized packages tailored to your needs, including Market Entry Research Package, Competitor Tracking Package, Supplier & Distributor Package and much more.
Disclaimer: Please note that the findings, conclusions and recommendations that TBRC Business Research Pvt Ltd delivers are based on information gathered in good faith from both primary and secondary sources, whose accuracy we are not always in a position to guarantee. As such TBRC Business Research Pvt Ltd can accept no liability whatever for actions taken based on any information that may subsequently prove to be incorrect. Analysis and findings included in TBRC reports and presentations are our estimates, opinions and are not intended as statements of fact or investment guidance.
Contact Us:
The Business Research Company
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email: info@tbrc.info
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company"
Oliver Guirdham
The Business Research Company
+44 7882 955267
info@tbrc.info
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.