Regenerative Medicine Market Set to Reach US$150.2 Bn by 2033, Says Persistence Market Research
The regenerative medicine market is rapidly evolving, driven by advances in cell and gene therapies, tissue engineering, and personalized healthcare solutions.
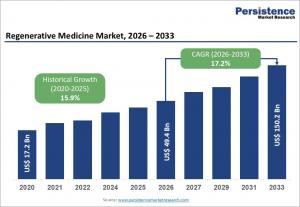
LONDON, UNITED KINGDOM, April 13, 2026 /EINPresswire.com/ -- The global regenerative medicine market is poised for remarkable growth, expanding from an estimated US$49.4 billion in 2026 to US$150.2 billion by 2033, reflecting a robust compound annual growth rate (CAGR) of 17.2% over the forecast period. This upward trajectory is fueled by advancements in digital healthcare, telehealth, and analytics, alongside increasing patient awareness and expanding healthcare infrastructure worldwide. North America continues to lead the market due to its established biotech ecosystem and stringent regulations that ensure high-quality production, while the Asia-Pacific region emerges as the fastest-growing market, driven by rising government initiatives, enhanced healthcare facilities, and significant investments in software, services, and interoperable manufacturing solutions.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/34638
Market Dynamics and Key Drivers
Advances in tissue engineering and biomaterials are central to the growth of regenerative medicine. Tissue engineering integrates living cells with scaffolds and biological signals to create constructs that mimic natural tissues such as skin, cartilage, and bone. Government-supported research programs, such as those funded by the National Institute of Dental and Craniofacial Research (NIDCR), emphasize engineering biocompatible scaffolds and smart materials that support growth and integration with native tissues. Biomaterials have evolved from passive supports to active agents that interact with cells to promote regeneration, including hydrogels, polymers, and bioactive matrices that enhance tissue repair and reduce reliance on donor tissues. Federal agencies like the National Institute of Standards and Technology (NIST) are developing measurement standards to ensure consistency and quality, accelerating the field’s adoption.
Challenges in Workforce and Infrastructure
Despite promising advancements, the regenerative medicine sector faces challenges related to workforce availability and specialized facilities. Skilled professionals in biology, cell culture, manufacturing, and quality assurance remain in short supply, limiting research throughput and slowing clinical development timelines. Industry surveys indicate that laboratories unable to hire experienced technical and production staff rose from 28.1% in 2018 to 35% in 2023. This shortage affects manufacturing, analytical development, and quality control roles, with recruitment timelines extending two to three months, highlighting the need for targeted training programs and expansion of specialized facilities to scale production efficiently.
Opportunities in Next-Generation Therapies
Next-generation stem cell and gene therapies represent significant growth opportunities. Global clinical trial databases reported 1,751 active regenerative medicine trials as of April 2024, including 442 cell therapy and 586 gene therapy trials targeting cardiovascular, neurological, and rare diseases. Regulatory milestones underscore this potential; in late 2025, the FDA approved Waskyra (etuvetidigene autotemcel) for Wiskott-Aldrich syndrome, while the NHS approved exa-cel, a CRISPR-based gene-edited stem cell therapy for sickle cell disease, achieving functional cures in over 96% of trial participants. These developments signal increasing regulatory flexibility and clinical validation for advanced therapies.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/34638
Product and Application Trends
Cell-based therapies dominate the regenerative medicine market, accounting for 49.7% of the global share in 2025. These therapies address fundamental mechanisms of tissue repair and functional restoration and are validated through over 1,700 active clinical trials worldwide. Applications in orthopedics and musculoskeletal care are particularly prominent due to high prevalence rates of musculoskeletal disorders affecting approximately 1.71 billion people globally. Techniques like stem cell therapy, scaffold-based tissue engineering, and Orthobiologics offer alternatives to surgery and prosthetics, supported by government research programs such as the NIH Musculoskeletal Tissue Engineering and Regenerative Medicine Program.
Regional Insights
North America holds a 43.5% market share in 2025, driven by robust research infrastructure, regulatory support, and high clinical activity, including 980 active regenerative medicine trials. The U.S. Food and Drug Administration’s RMAT designation streamlines development and review, enhancing commercial adoption. Europe follows closely, with supportive funding, collaborative research networks, and regulatory approvals, including over 25 Advanced Therapy Medicinal Products (ATMPs) endorsed by the European Medicines Agency. Asia-Pacific is the fastest-growing region, with over 500 active clinical trials in Japan, China, and South Korea, supported by favorable regulations and a rapidly ageing population exceeding 700 million, driving demand for regenerative solutions.
Competitive Landscape
Leading companies prioritize scalable manufacturing, regulatory compliance, and advanced cell and gene therapy technologies. Strategic collaborations with biotech firms, academia, and regulators accelerate adoption, while investments in AI-enabled design and high-throughput testing ensure product reproducibility and efficacy. Notable developments include Novartis receiving FDA Breakthrough Therapy designation for ianalumab in Sjögren’s disease and Roche acquiring Poseida Therapeutics’ cell therapy portfolio. Key players include Pfizer, Novartis, Roche, Bayer, AstraZeneca, GlaxoSmithKline, Merck KGaA, Abbott, Integra LifeSciences, Vericel, Cook Biotech, and Astellas Pharma.
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/34638
Market Segmentation
By Product Type
Cell-Based Therapies
Gene Therapies
Tissue Engineering
Others
By Application
Orthopedics & Musculoskeletal
Oncology
Cardiovascular
Dermatology & Wound Healing
Others
By End-user
Hospitals & Clinics
Research Institutes & Academic Centers
Pharmaceutical & Biotechnology Companies
Others
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East and Africa
The regenerative medicine market’s trajectory reflects a convergence of technological innovation, clinical advancement, and strategic investment, signaling transformative opportunities across global healthcare. With ongoing breakthroughs in cell and gene therapies, the market is set to redefine treatment paradigms and deliver personalized, effective solutions for patients worldwide.
Read Related Reports:
Controlled Substance API Market: The global controlled substance API market is valued at US$14.9 billion in 2026 and projected to reach US$27.9 billion by 2033, growing at a 9.4% CAGR.
Viscosupplementation Market: Global viscosupplementation market to grow from US$3.4 Bn in 2026 to US$5.2 Bn by 2033, expanding at a CAGR of 6.2% during the forecast period.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.